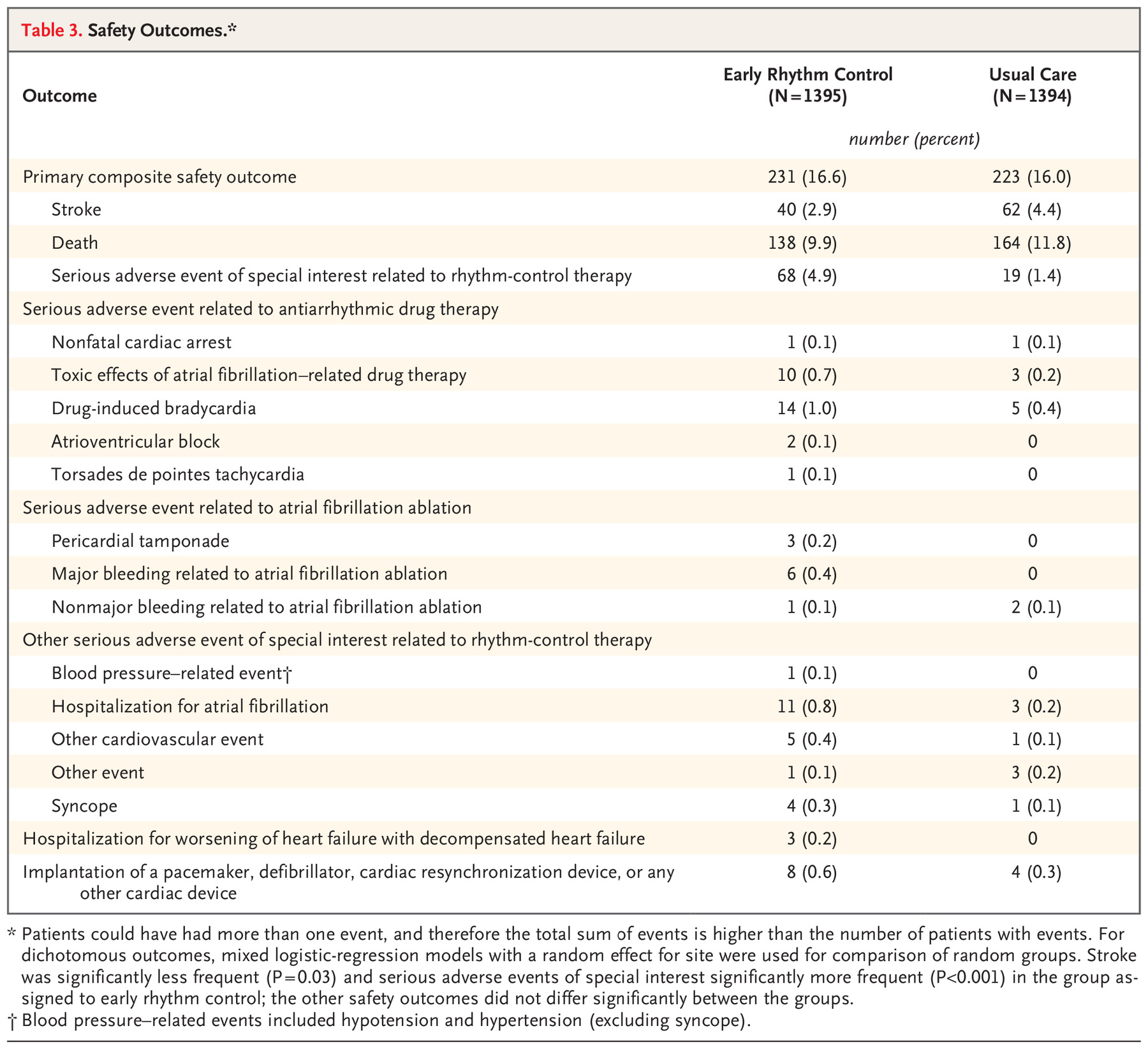
Early Rhythm-Control Therapy in Patients with Atrial Fibrillation
In this international, investigator-initiated, parallel-group, open,
blinded-outcome-assessment trial, the investigators randomly assigned patients
who had early atrial fibrillation (diagnosed ≤1 year before enrollment)
and cardiovascular conditions to receive either early rhythm control or usual care.
N = 2789, mean age 70, 46% female, f/u 5.1yrs.
The first primary outcome was a composite of death from cardiovascular causes, stroke, or hospitalization with worsening of heart failure or acute coronary syndrome.
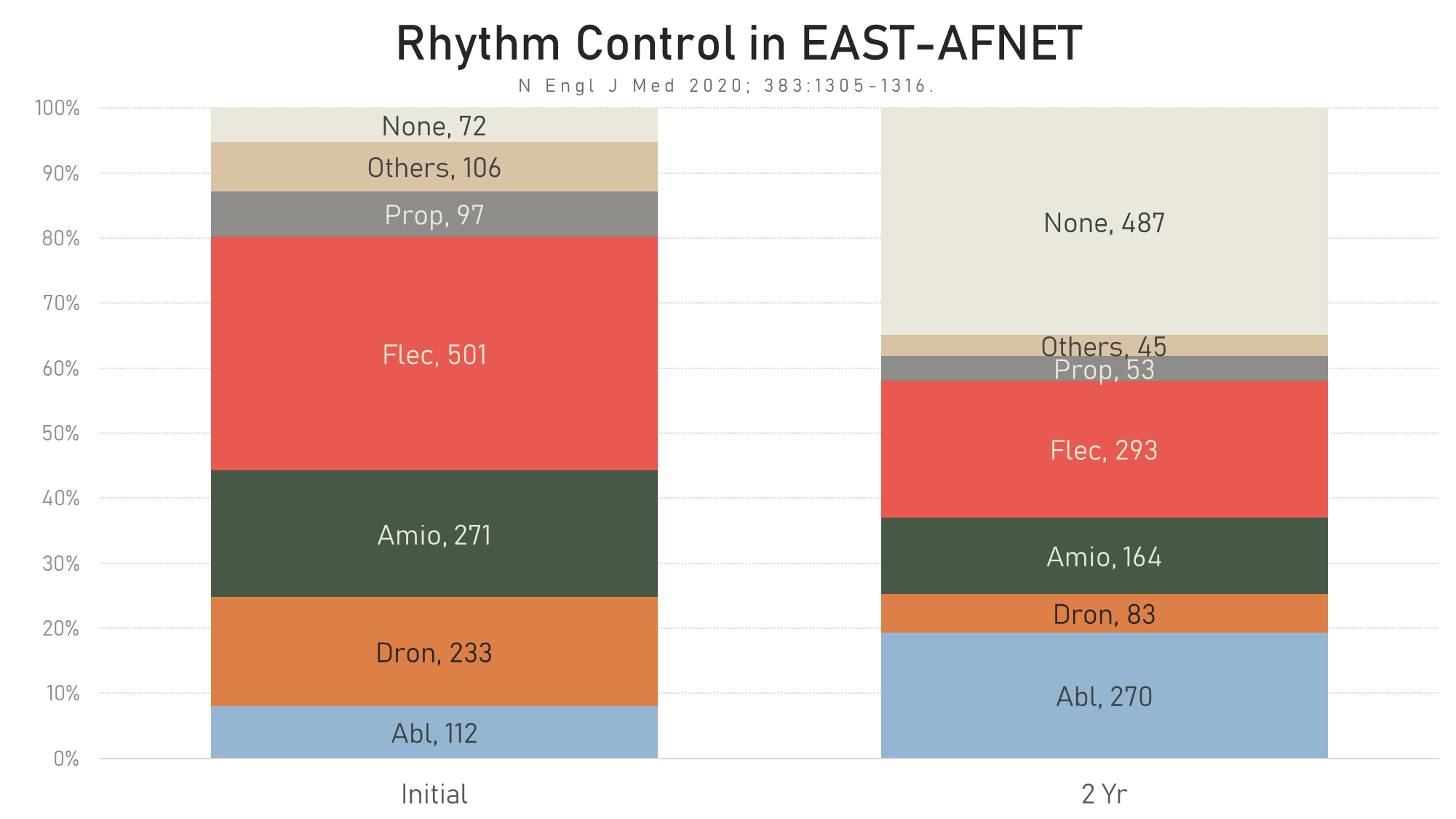
Type of rhythm control.
Prop = Propafenone, Flec = Flecainide, Amio = Amiodarone, Dron = Dronedarone, Abl = Ablation
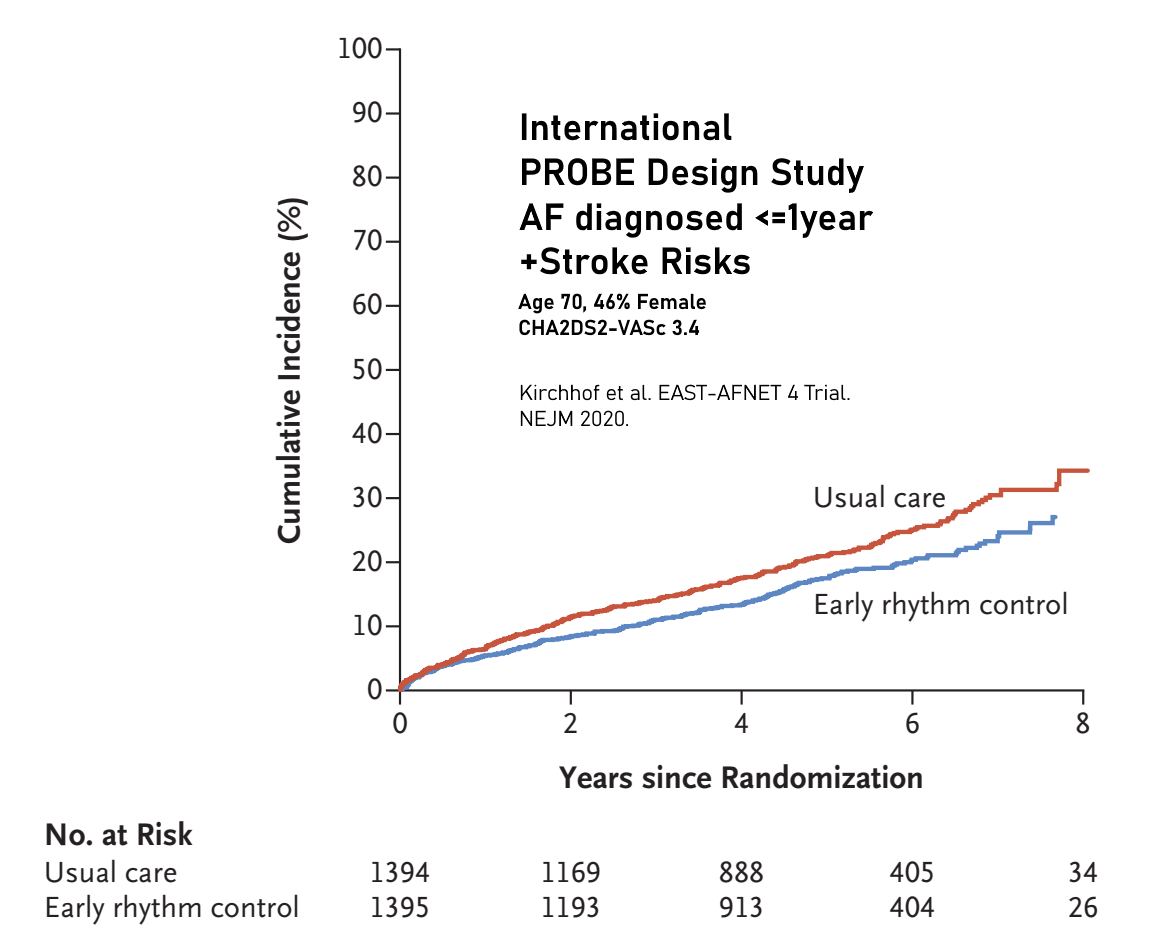
Cumulative Incidence for the First Primary Outcome.
HR (early rhythm control vs. usual care), 0.79; 96%CI, 0.66 to 0.94; P=0.005.
The trial was stopped for efficacy at the third interim analysis after
a median follow-up of 5.1 years per patient.
A first-primary-outcome event occurred in 249 patients assigned to receive
early rhythm control (3.9 per 100 person-years) and in 316 patients assigned
to receive usual care (5.0 per 100 person-years).
In AFFIRM, rhythm control was not associated with better CV outcomes than rate control.
Rhythm control was on the other hand associated with higher non-CV death than that of rate control,
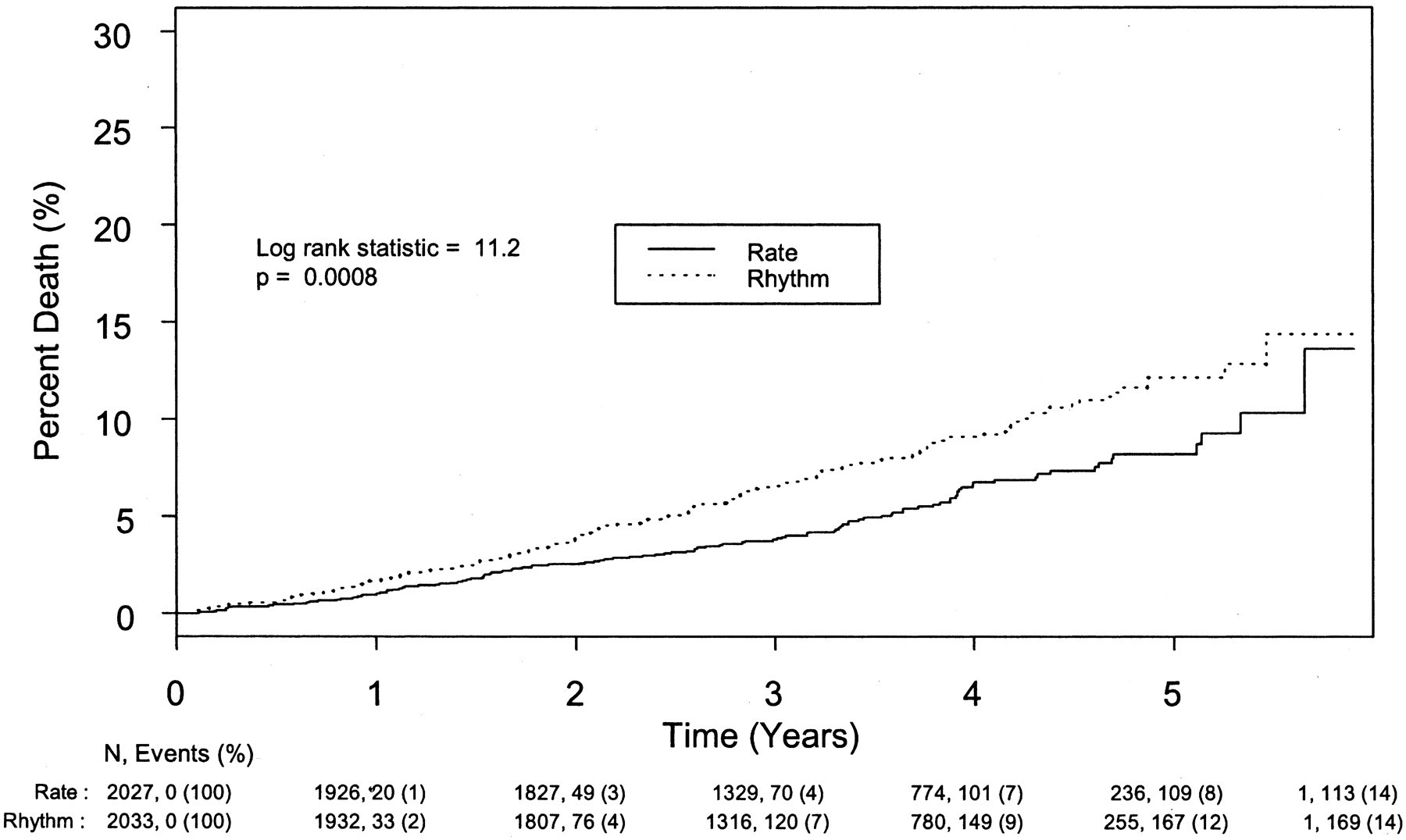
Cumulative noncardiovascular mortality in the rhythm-control and rate-control groups in AFFIRM.
The type of rhythm control treatment in AFFIRM vs. EAST-AFNET4 is shown here.
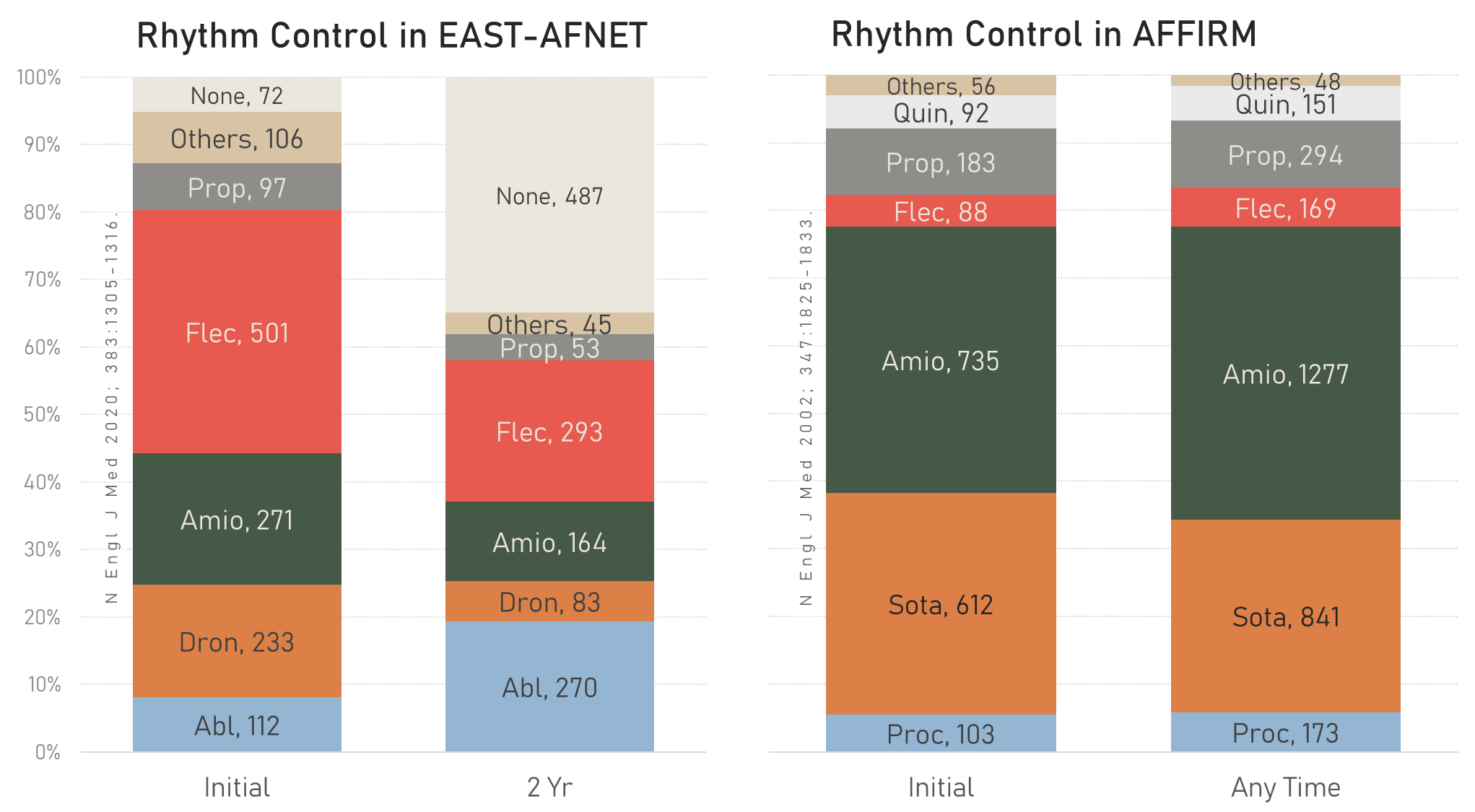
Type of rhythm control EAST-AFNET4 vs. AFFIRM.
Prop = Propafenone, Flec = Flecainide, Amio = Amiodarone, Dron = Dronedarone, Abl = Ablation,
Quin = Quinidine, Sota = Sotalol, Proc = Procainamide.
The CV events in both groups were relatively low. In both group, the incidence of stroke was less than 1 in 100 person-year
and the all-cause mortality was less than 1.5 per 100 person-year.
The adverse events were also low, though slightly higher in rhythm control group.